Cell Biology Laboratory
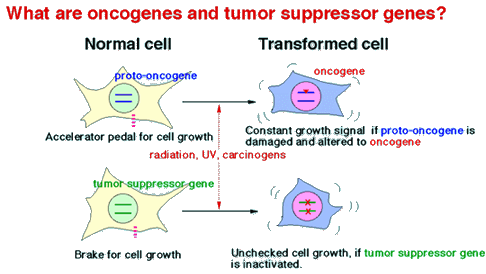
Figure: Molecular Oncology/Epidemiology Studies
The body consists of a variety of cells, which are differentiated from a fertilized cell after numerous divisions. Both genetics and the environment influence somatic cell division, resulting in active growth, division arrest, or apoptosis (cell suicide). Proto-oncogenes and tumor suppressor genes act as the accelerators and brake pedals, respectively, in cell growth. If these genes are damaged, the fine balance is disrupted, and growth dysregulation ensues, possibly resulting in the formation of malignant tumor cells.
Among tumors, the mutation spectra in oncogenes and/or tumor suppressor genes are quite heterogeneous. At present, we are using state-of-the-art methods to identify tissue-specific genetic damage caused by atomic bomb radiation. Moreover, because the amount of tissue materials is severely limited, we are also investigating ways of improving detection techniques.
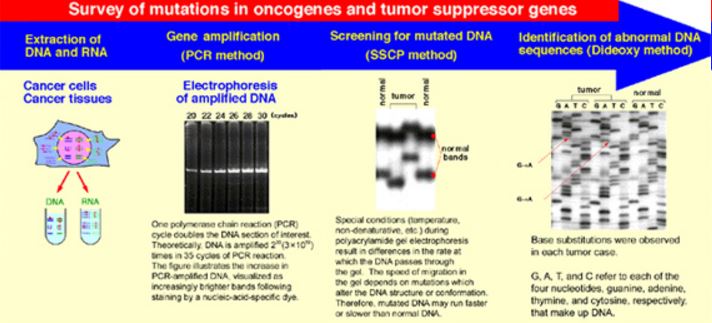
Figure: Molecular Oncology/Epidemiology Studies
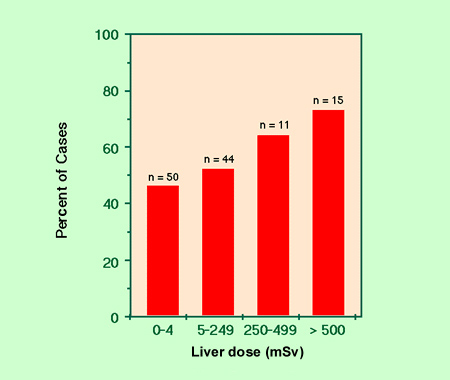
Figure 1. Frequency of cases with P53 mutation in hepatocellular carcinomas from the atomic-bomb survivors
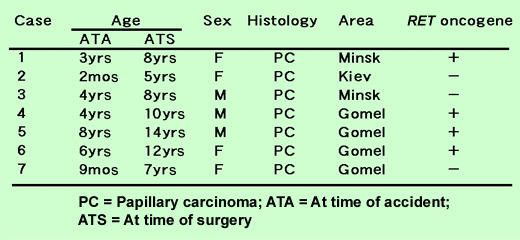
Table 1. RET oncogene activation in thyroid cancer in children from areas contaminated by the Chernobyl accident
Conclusion
Environmental factors, such as radiation and/or chemical mutagens, are well known to induce many mutations in genomic DNA. Such mutations, successively induced in various oncogenes and tumor suppressor genes, may transform normal cells into malignant ones.
A dose-dependent increase in point mutations within the tumor suppressor gene p53 was observed in liver cancer cells of atomic-bomb survivors (Figure 1). In another recent study, we demonstrated that the RET proto-oncogene frequently rearranged to the RET/PTC1 ongocene in childhood thyroid cancers from areas contaminated by the Chernobyl nuclear power plant accident (Table 1).
These facts suggest that radiation damage to cancer-related genes may explain the increased incidence of cancer among the exposed in these studies. In the future, continued molecular oncological studies may shed light on whether such gene alterations are radiation-induced in the atomic-bomb survivors and what mechanisms are operating in radiation-induced carcinogenesis.

